Spec′tro-scope.
An instrument for viewing the spectra formed by the solar rays and by those of other bodies in an incandescent condition.
 |
|
Steinheil's spectroscope and spectrum. |
Newton, 1675, determined the fact that the solar light was composed of six different and unequally refrangible colors.
(See prism.) He also found that these colors, which combined to form white light, could not be farther separated by undergoing a second refraction In 1781, Scheele, who discovered the fact that light exercises a chemical action upon chloride of silver, found that the compound was blackened most toward the violet
[
2259]
end of the spectrum.
Subsequent researches have shown that the solar spectrum is apparently composed of three parts, which are. throughout a portion of their extent, superimposed one upon the other (
B.
Fig. 5357); thus the part
h exhibits the maximum
heating effect; the part
l is that which produces light; and the part
e exerts the greatest
chemical or actinic action.
The curves indicate the respective maxima and minima of heat, light, and actinic force respectively; the first being greatest at a point entirely without the visible end of the spectrum; the second commencing just beyond the line
A near the end of the red, and extending beyond the line
H in the violet; and the third commencing near the central part of the visible spectrum, increasing to a maximum near the violet end, and only disappearing at a point far beyond the visible part of the spectrum.
At the beginning of this century
Dr. Wollaston, in repeating the Newtonian experiment, admitted a beam of light through a very narrow slit instead of a round hole, and detected a number of black lines in the spectrum; and in 1814, Fraunhofer, pursuing the investigation, had discovered and located 576 of these lines.
He also observed that these were uniformly the same in light received directly from the sun and reflected from the planets, and that light from the selfluminous fixed stars contained black lines differing from those of the solar light.
He thence concluded that these variations were due to causes existing outside of our atmosphere,—a conclusion which has been since amply confirmed.
Melville, in 1752, noticed the yellow flame due to sodia; and in 1822
Sir John Herschel remarked that “the colors contributed by different objects to flame afford in many instances a ready and neat way of detecting extremely minute quantities of them.”
Mr. Fox Talbot, in 1834.
distinguishes the difference between the red lines produced by the flames of strontia and lithia, and in 1845
Professor W. A. Miller experimented upon the spectra of the alkaline earth metals.
Professor Bunsen, however, so far advanced the subject that he, in conjunction with
Kirchoff, may be almost said to have invented spectrum analysis as it now exists.
Through its instrumentality
Bunsen discovered, in 1860, the new metals caesium and rubidium;
Crookes, in 1861, discovered thallium; and in 1864
Reich and
Richter discovered indium.
In
Fig. 5357,
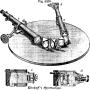
A represents the improved spectroscope of Steinheil.
It consists of a stand
c carrying the flint-glass prism
a, having a refracting angle of 60°. The stand has two arms, one of which carries the telescope
f, and the other the tube
g, containing a lens in the end nearest the prism, and at the other end a scale which can be seen through the telescope by reflection from the surface of the prism.
The light
e is admitted to the tube
f through an adjustable slit in the piece
d, the upper part only of which is open; the light
e′ is reflected from a prism within the piece
d below the slit, so that the spectra of the two lights appear one above the other in the telescope
f, the scale in the tube
g being illuminated when required by a lamp in front of it; the adjustments consist in properly focusing the telescope and so arranging the tubes
b g and the lights that their spectra and the micrometer scale are all distinct in the same field.
In
Fig. 5358, the arrangement used by
Kirchoff, a train of four prisms is employed; the rays passing through the slit in
d are rendered parallel by a lens in the tube
a, and, being decomposed successively by the prisms, are, on emerging from that at the left, viewed by the telescope
b. This arrangement gives a very elongated spectrum
c d represent two forms of the piece by which the upper beam is transmitted and the lower reflected through the tube
a. The width of the slit may be varied by means of screws.
 |
|
Spectroscopic apparatus and battery. |
Fig. 5359 shows the apparatus employed for volatilizing the metals in order to obtain their spectra; it consists essentially of a voltaic battery
a, induction coil
b, and a stand
c having adjustable arms, carrying the points between which the substance
d is held while being subjected to the action of the current.
The luminous image of the burning metal is transmitted through a slit and tube to the prism and viewed by a telescope.
Faraday ascertained that the electric spark is but the effect of the combustion of minute particles of the terminals and the air or other matter through which it passes from one to the other.
The spectroscope has already given us an insight of the elements present in the sun and some of the fixed stars, and of the nature of the components of other heavenly bodies, as the nebulae and comets.
It has also been employed to determine the exact moment proper for adding the spiegeleisen and for stopping the conversion in the Bessemer steel process.
See pages 277, 613.
It is as yet comparatively in its infancy, and will no doubt some day be of great service in practical metallurgy, as it has already been in qualitative chemical analysis.
Mr. Norman Lockyer has successfully used the spectroscope as a means of quantitative analysis for testing alloys in the
British mint.
His apparatus consists of an electric lamp, in the lower carbon of which a recess is made to receive the alloy to be vaporized.
The spectrum is thrown on a screen in a closed box and is photographed, admitting of ready comparison with the previously photographed spectra of other alloys containing known proportions of the metals.
By means of a delicate scale attached to the instrument, the coincidence or variation of the lines in the different spectra, arranged one above the other, is determined, the relative length, strength, and position of these lines indicating the proportion of each metal contained in the alloy.
Huggins has applied spectroscopic observation to the determining of the proper motion of the heavenly bodies, by observing the displacement of the spectral lines.